Life sciences teams don’t fail at regulatory submissions because they don’t understand eCTD.
They fail because everything that happens before and around eCTD is chaotic, misaligned, and poorly governed.
Govern 365 was built to fix that exact gap.
To understand why, you first need to understand one uncomfortable truth:
eCTD is not a collaboration system — and it was never meant to be.
Yet teams keep forcing collaboration, review, and partner sharing into places where it doesn’t belong. That’s where risk enters. And that’s precisely where Govern 365 fits.
The Real Problem Isn’t eCTD — It’s Everything Around It
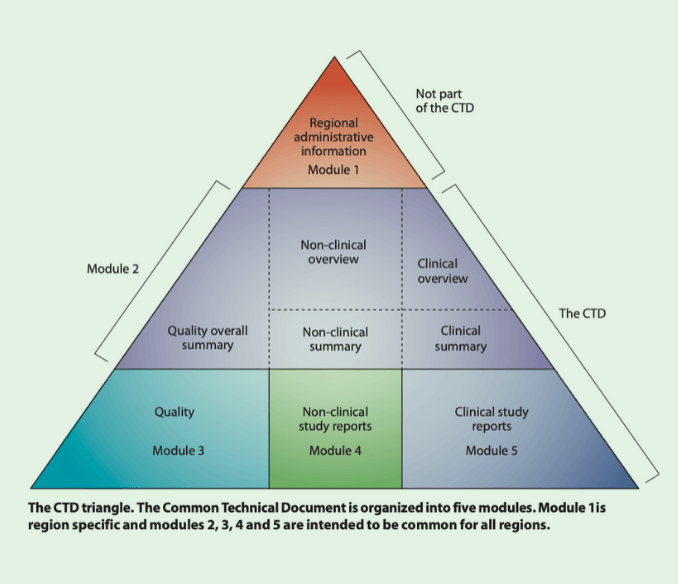
eCTD (electronic Common Technical Document) is a globally standardized, XML‑driven submission and lifecycle system used to deliver regulatory dossiers to authorities like the FDA, EMA, PMDA, and others.
It is excellent at what it does:
- Managing submission sequences
- Tracking lifecycle operations (new, replace, append, delete)
- Preserving immutable regulatory history
- Enabling structured regulator review
But eCTD was never designed for:
- Drafting
- Cross‑functional collaboration
- CRO and partner review
- Executive visibility
- Inspections outside formal submissions
Still, this is exactly where most teams struggle — long before the first eCTD sequence even exists.
Where Most Teams Go Wrong
A common misconception sounds like this:
“We already have CTD‑like folders, so we’re eCTD ready.”
That assumption creates three dangerous behaviors:
- Treating folders like lifecycle systems
- Treating VDRs like submission platforms
- Treating SharePoint like a regulatory system of record
None of these are true — and each introduces compliance risk.
Why Traditional VDRs Fail in eCTD Workflows
Most legacy VDRs were built for deals, not regulatory lifecycles.
They typically:
- Mimic CTD folder structures without understanding eCTD rules
- Allow uncontrolled versioning
- Blur draft, approved, and submitted states
- Encourage manual uploads and exports
- Sit outside the organization’s core security perimeter
The result?
- Confusion during inspections
- Broken traceability
- Risky handoffs into eCTD publishing tools
- Teams quietly using VDRs as if they were submission systems
This is exactly what regulators do not want.
eCTD, DMS, and VDR Are Not the Same Thing
These systems serve different moments in the regulatory lifecycle:
| System | What It’s For |
| DMS | Authoring, drafting, internal collaboration |
| VDR | Secure external sharing, structured review, inspections |
| eCTD | Regulatory submission, XML lifecycle control |
Trying to collapse these roles into a single platform usually creates process shortcuts — and audit exposure.
This Is the Gap Govern 365 Was Designed to Fill
Govern 365 was not built to replace eCTD.
It was built to respect eCTD.
Specifically, Govern 365 acts as the regulatory‑aware VDR layer that sits:
- Upstream of eCTD (during drafting, review, and approval)
- Side‑by‑side post‑submission (for inspection and reference)
- Never inside the eCTD system itself
This separation is not a limitation — it is by design.
Download “Unlocking Growth in Life Sciences with Govern 365” Ebook
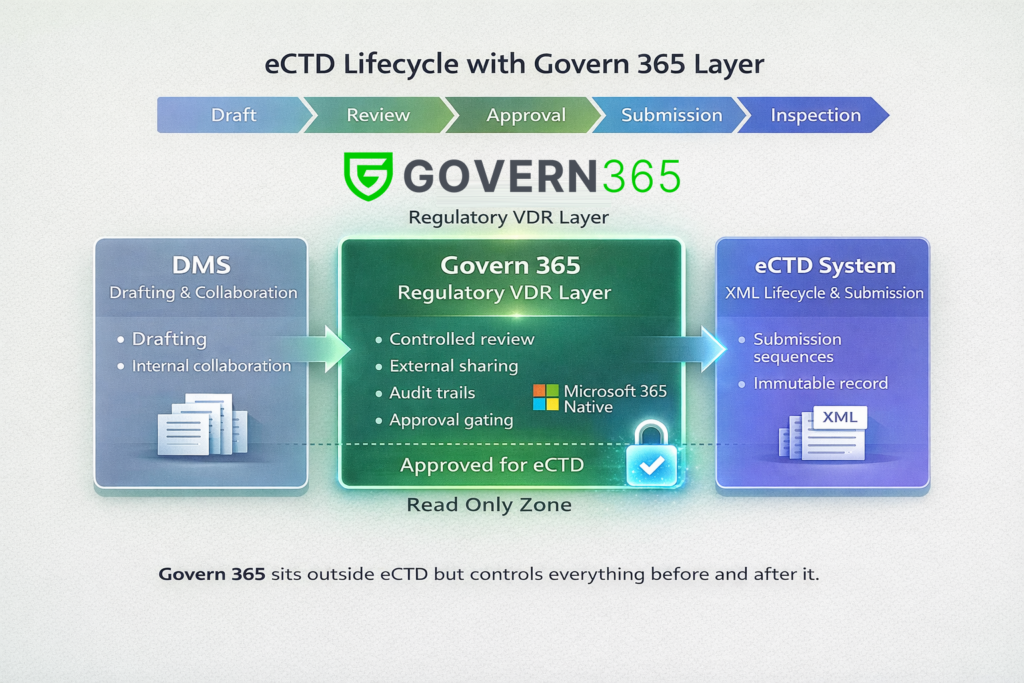
The eCTD Lifecycle — With Govern 365 in the Right Place
Phase 1: Drafting and Pre‑Submission Reality
- Documents are fluid
- CROs, clinicians, QA, and legal teams collaborate
- No submission sequence exists yet
Govern 365 role
- Optional but powerful
- Secure external sharing
- Early diligence collections
- Controlled reviewer access
No CTD enforcement yet — and that’s correct.
Phase 2: Cross‑Functional Review (Where Govern 365 Becomes Critical)
As content stabilizes:
- Medical, QA, Legal, and Regulatory review intensifies
- Approval workflows begin
- Inspection risk starts to matter
Govern 365 enables
- Role‑based, time‑bound access
- Controlled commenting and Q&A
- Review traceability and audit logs
- CTD‑aligned structure without pretending to be eCTD
This makes reviewers feel “submission ready” — without introducing XML complexity.
Phase 3: Approval and Lock — The Most Important Boundary
This is where most teams make mistakes.
Govern 365 introduces a clear, enforceable handoff point:
Approved‑for‑eCTD Area
- Read‑only
- Metadata complete (module, section, region)
- No collaboration features
- No version churn
✅ This is the only place eCTD publishers should pull content from.
This single boundary dramatically reduces submission and inspection risk.
Phase 4: eCTD Publishing (Govern 365 Steps Out)
Once Regulatory Ops begins publishing:
- XML backbone is generated
- Lifecycle operations are assigned
- Technical validation is run
At this point:
- The eCTD tool becomes the system of record
- Govern 365 does not interfere
- No mirroring, syncing, or pretending
This separation is compliance‑critical.
We’re Trusted by the Best


Govern 365 is a strong, Microsoft 365 native governance and secure collaboration platform. Overall, it comes close to becoming a contender in the governance and secure collaboration market.
Christopher Dixon
Senior IT Director

Phase 5: Submission, Review, and Inspections
After submission:
- Regulators review eCTD sequences
- Questions and amendments follow
- Executives and partners still need visibility
Govern 365 re‑enters safely
- Read‑only submitted reference
- Inspection‑ready access
- No lifecycle manipulation
- No risk to the regulatory record
Why Govern 365 Is Different from “Just SharePoint”
Govern 365 is Microsoft 365‑native, but it is not “just SharePoint sharing”.
It adds:
- Regulatory‑aware structure
- Granular external access controls
- Full auditability
- Approval gating
- Clear lifecycle boundaries
All inside your Microsoft 365 tenant, governed by your own security and compliance policies.
No third‑party data silos.
No per‑page fees.
No black‑box regulatory risk.
The Key Takeaway
eCTD is not a folder problem.
It is a lifecycle problem.
Govern 365 exists because:
- Collaboration must happen before eCTD
- Visibility must exist after submission
- And neither should ever contaminate the eCTD system itself
That’s how clean architectures are built.
That’s how inspections become boring.
And that’s why Govern 365 exists.
Want to see how Govern 365 fits cleanly into your eCTD workflow?